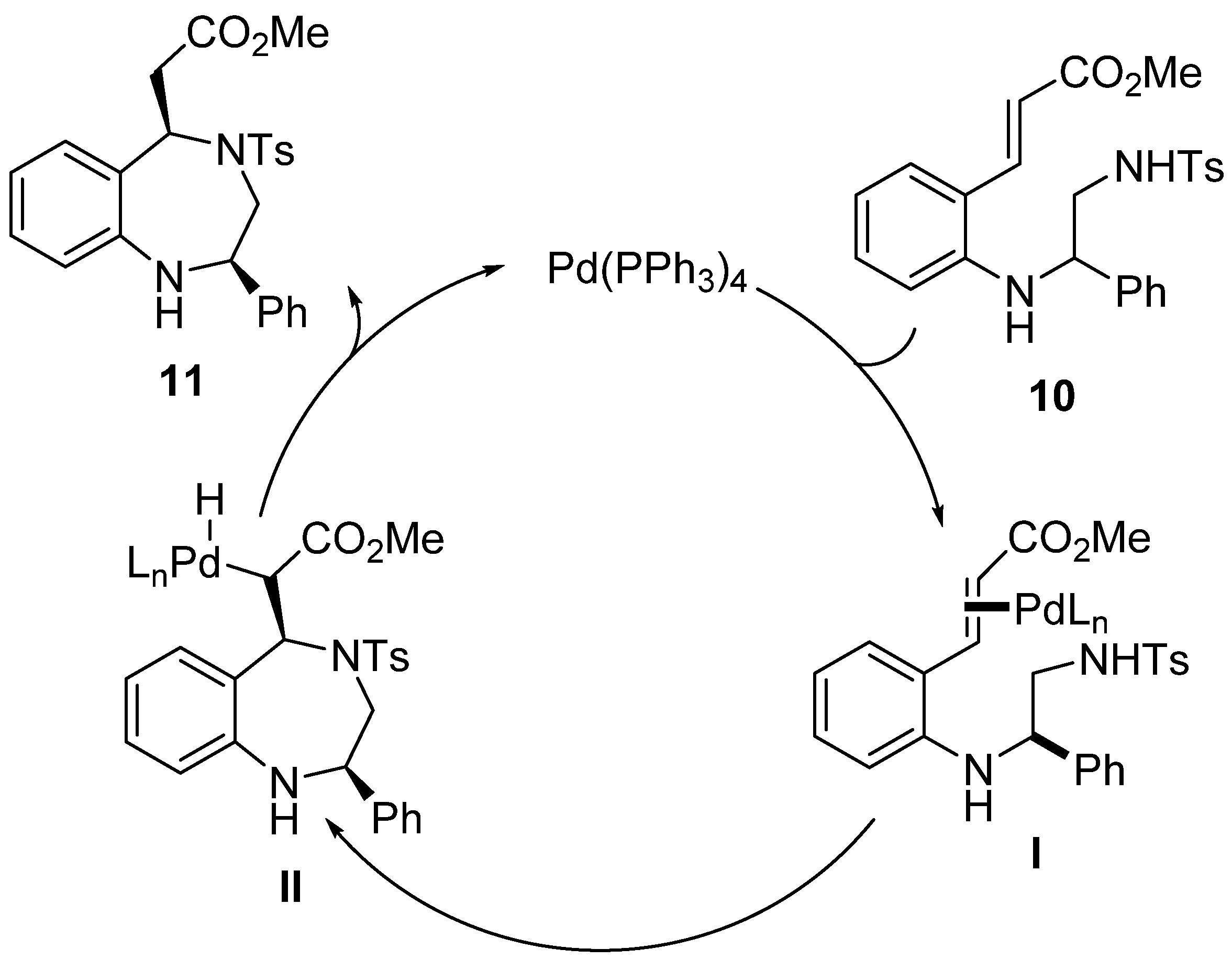
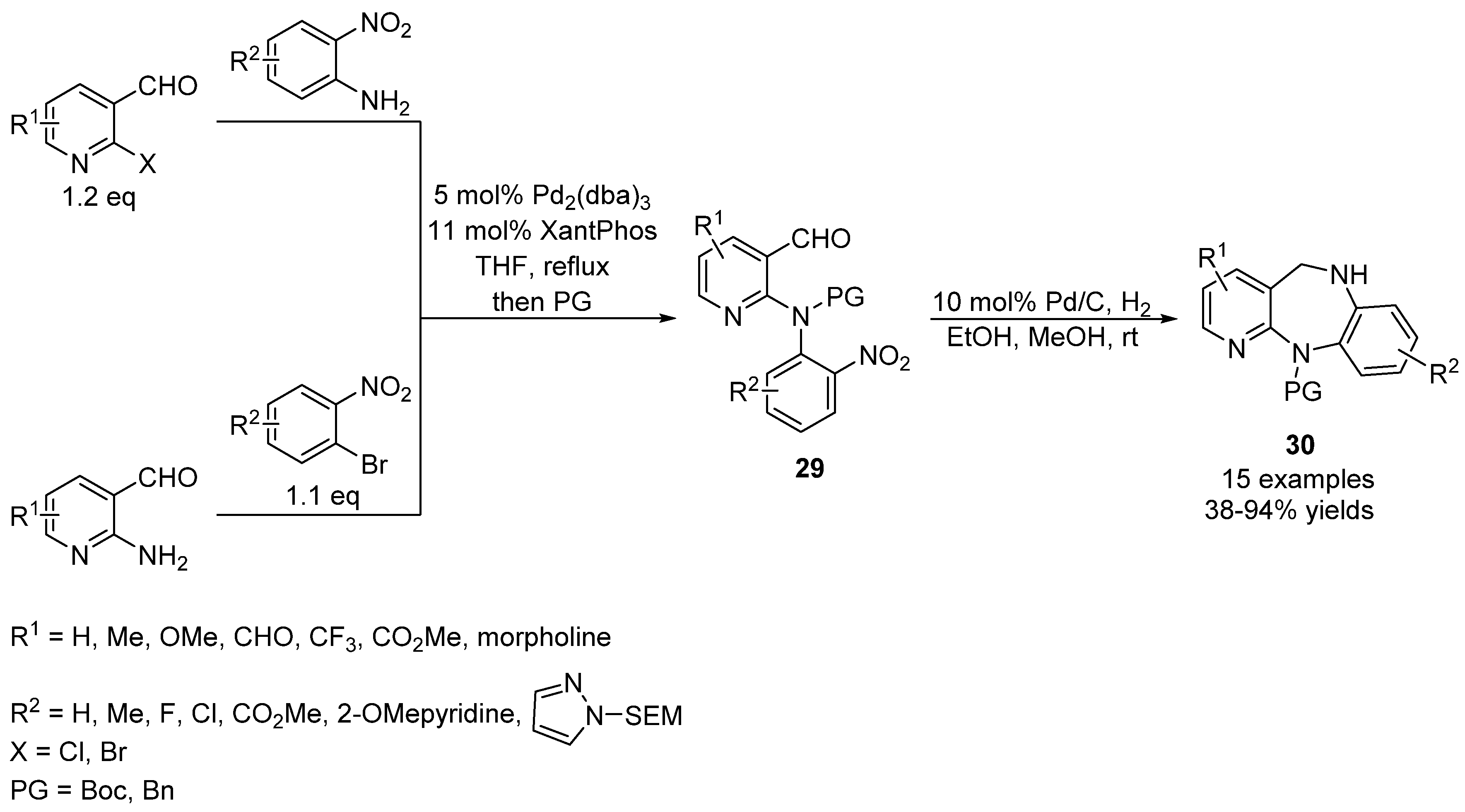
Catalysts | Free Full-Text | Diaryliodoniums Salts as Coupling Partners for Transition-Metal Catalyzed C- and N-Arylation of Heteroarenes | HTML

Synthesis of Biaryls via Decarbonylative Palladium-Catalyzed Suzuki-Miyaura Cross-Coupling of Carboxylic Acids - ScienceDirect
PDF) Synthesis of Biaryls via Decarbonylative Palladium-Catalyzed Suzuki-Miyaura Cross-Coupling of Carboxylic Acids

Cross‐Coupling of Aryl/Alkenyl Pivalates with Organozinc Reagents through Nickel‐Catalyzed CO Bond Activation under Mild Reaction Conditions - Li - 2008 - Angewandte Chemie International Edition - Wiley Online Library

Synthesis of Biaryls via Decarbonylative Palladium-Catalyzed Suzuki-Miyaura Cross-Coupling of Carboxylic Acids - ScienceDirect

Selective PdII‐Catalyzed Acylation of Pyrrole with Aldehydes. Application to the Synthesis of Celastramycin Analogues and Tolmetin - Santiago - 2020 - European Journal of Organic Chemistry - Wiley Online Library

In Situ Generation of Palladium Nanoparticles: Ligand-Free Palladium Catalyzed Pivalic Acid Assisted Carbonylative Suzuki Reactions at Ambient Conditions
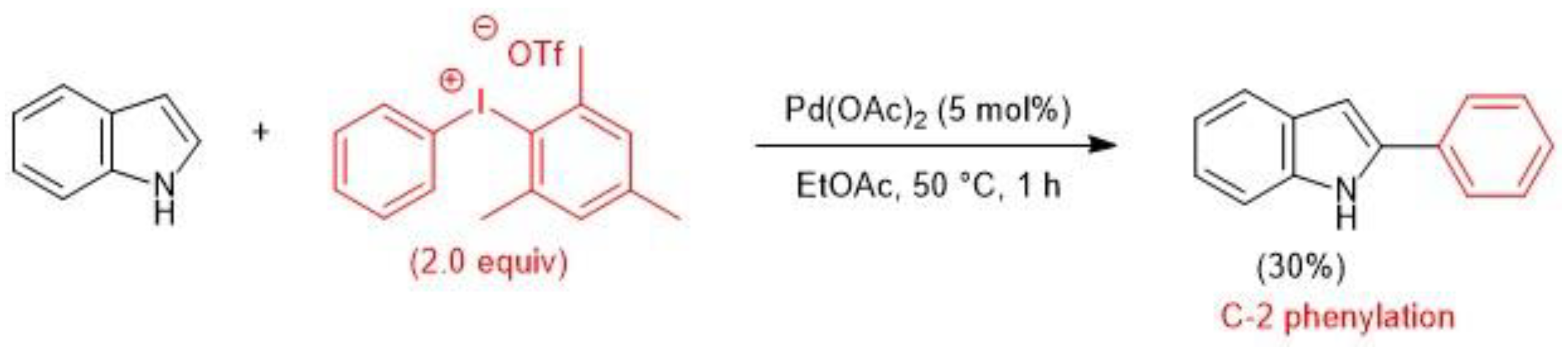
Catalysts | Free Full-Text | Diaryliodoniums Salts as Coupling Partners for Transition-Metal Catalyzed C- and N-Arylation of Heteroarenes | HTML

Recent Applications of Palladium‐Catalyzed Coupling Reactions in the Pharmaceutical, Agrochemical, and Fine Chemical Industries - Torborg - 2009 - Advanced Synthesis & Catalysis - Wiley Online Library




![A [13]rotaxane assembled via a palladium molecular capsule | Nature Communications A [13]rotaxane assembled via a palladium molecular capsule | Nature Communications](https://media.springernature.com/m685/springer-static/image/art%3A10.1038%2Fs41467-019-11635-6/MediaObjects/41467_2019_11635_Fig1_HTML.png)