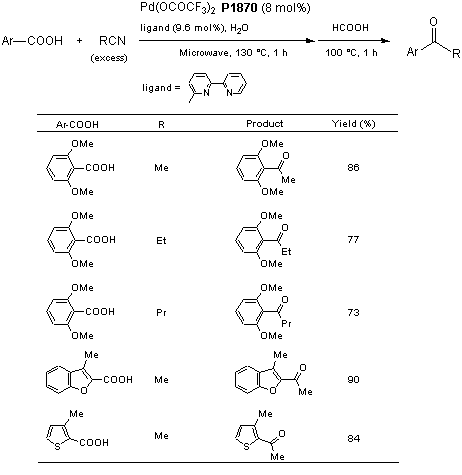
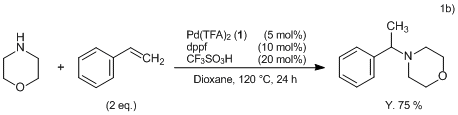
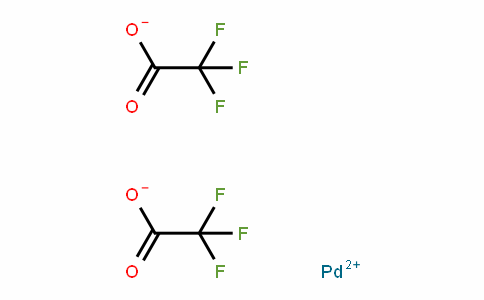
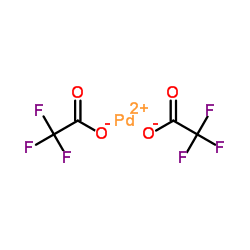
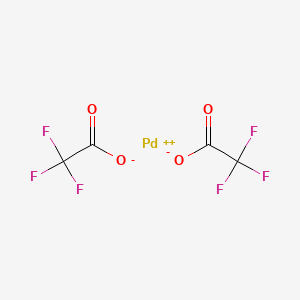
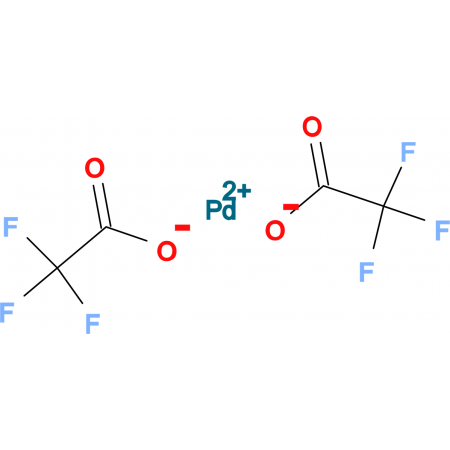
Formation of anionic palladium(0) complexes ligated by the trifluoroacetate ion and their reactivity in oxidative addition - ScienceDirect

Palladium(II) trifluoroacetate, 97%, ACROS Organics™ 5g; Glass bottle Palladium(II) trifluoroacetate, 97%, ACROS Organics™ | Fisher Scientific

China Palladium(II) Trifluoroacetate (Pd(TFA)2) CAS No.: 42196-31-6 Manufacturers - Free Sample - Alfa Chemical
Palladium(ii) Trifluoroacetate,97% Pd(cf3coo)2cas No.42196-31-6 - Buy 42196 - 31 - 6 Product on Alibaba.com
![42196-31-6・Palladium(II) Trifluoroacetate・167-27681・163-27683[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation 42196-31-6・Palladium(II) Trifluoroacetate・167-27681・163-27683[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/sc/05/163-27683.png)
42196-31-6・Palladium(II) Trifluoroacetate・167-27681・163-27683[Detail Information] | [Synthesis & Materials] |Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation

China Palladium(II) Trifluoroacetate (Pd(TFA)2) CAS No.: 42196-31-6 Manufacturers - Free Sample - Alfa Chemical
1-cyclohexatrienyl-N,N-dimethyl-methanamine; palladium(+2) cation; tricyclohexylphosphane; 2,2,2-trifluoroacetate | C29H45F3NO2PPd | ChemSpider

PDF) Dehydrogenation of cyclohexanones catalyzed by palladium(II) trifluoroacetate | Jacques Muzart - Academia.edu
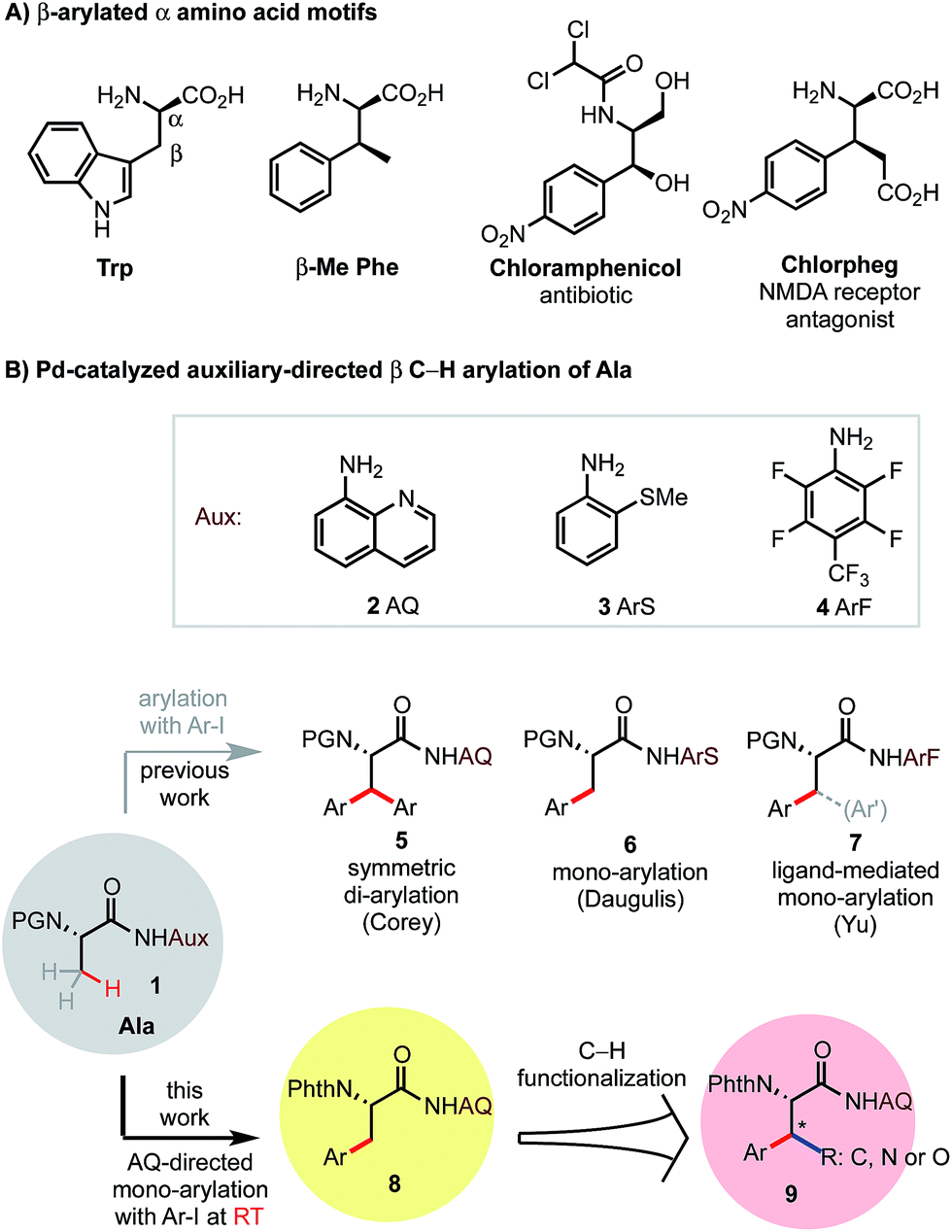
Palladium-catalyzed trifluoroacetate-promoted mono-arylation of the β-methyl group of alanine at room temperature: synthesis of β-arylated α-amino acids through sequential C–H functionalization - Chemical Science (RSC Publishing)

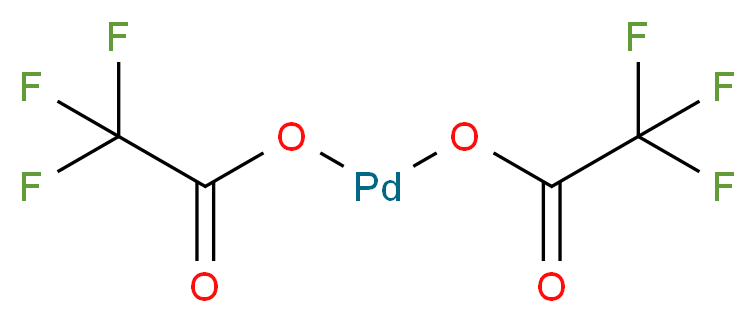
42196-31-6 | Palladium(II) Trifluoroacetate | Palladium Bis(2,2,2- trifluoroacetate); Palladium Bis(trifluoroacetate); Palladium Trifluoroacetate;Trifluoroacetic Acid palladium(2+) Salt Trifluoroacetic Acid Palladium(II) Salt; | C₂HF₃O₂ • 1/2(Pd) | TRC
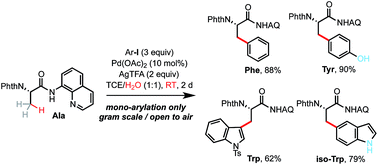
Palladium-catalyzed trifluoroacetate-promoted mono-arylation of the β-methyl group of alanine at room temperature: synthesis of β-arylated α-amino acids through sequential C–H functionalization - Chemical Science (RSC Publishing)

Palladium(II) Trifluoroacetate: An Efficient Pd-Catalyst for Coupling Reactions | Tokyo Chemical Industry Co., Ltd.(APAC)

%20Trifluoroacetate%20Powder.png)