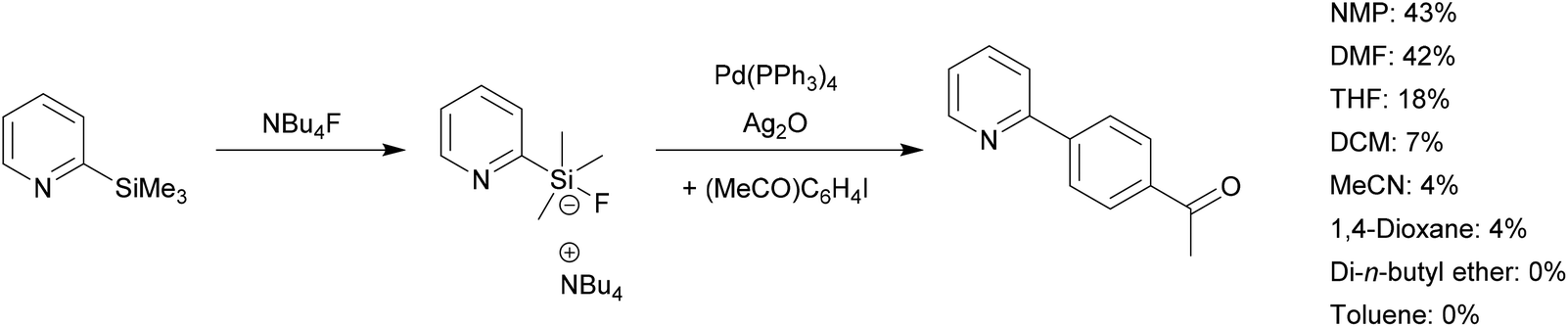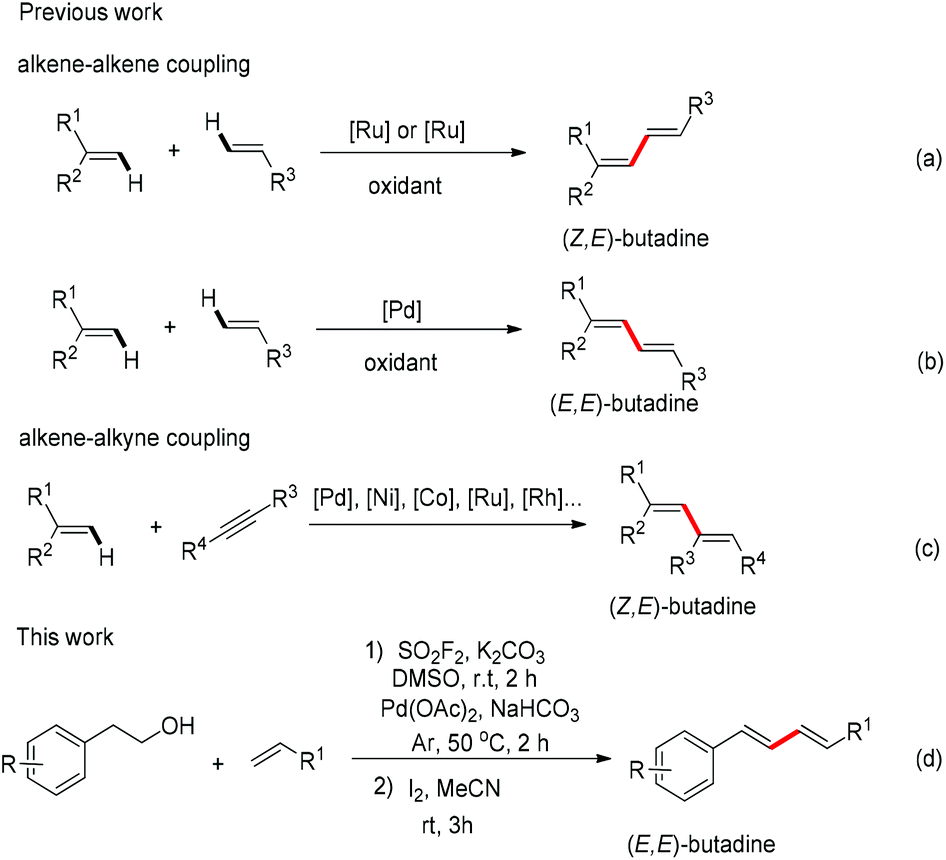
SO 2 F 2 mediated dehydrative cross-coupling of alcohols with electron-deficient olefins in DMSO using a Pd-catalyst: one-pot transformation of alcoho ... - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C8QO01388H

A ligand-free Pd(OAc)2 catalyst for the Wacker oxidation of styrene derivatives using hydrogen peroxide as the oxidant - ScienceDirect
Aqueous-Phase Chemistry of η3-Allylpalladium(II) Complexes with Sulfonated N-Heterocyclic Carbene Ligands: Solvent Effects in t

Figure 1 from On the mechanism of the palladium(II)-catalyzed decarboxylative olefination of arene carboxylic acids. Crystallographic characterization of non-phosphine palladium(II) intermediates and observation of their stepwise transformation in Heck ...
Catalysts | Free Full-Text | The Reims Journey Towards Discovery and Understanding of Pd-Catalyzed Oxidations | HTML

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
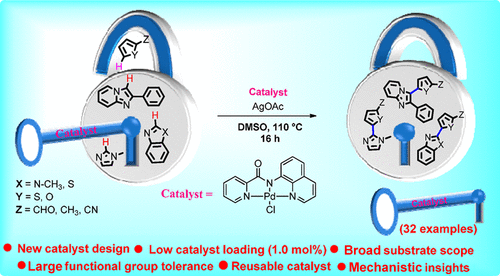
![Regioselective synthesis of 2,3-disubstituted 1-alkyl pyrrolo[2,3-b] quinoxalines through palladium-catalyzed Heck reaction of chalcones and evaluation of their anti-bacterial activities - ScienceDirect Regioselective synthesis of 2,3-disubstituted 1-alkyl pyrrolo[2,3-b] quinoxalines through palladium-catalyzed Heck reaction of chalcones and evaluation of their anti-bacterial activities - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0040402018303284-fx1.jpg)
Regioselective synthesis of 2,3-disubstituted 1-alkyl pyrrolo[2,3-b] quinoxalines through palladium-catalyzed Heck reaction of chalcones and evaluation of their anti-bacterial activities - ScienceDirect

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Table 2 | Cellulose nanocrystal supported palladium as a novel recyclable catalyst for Ullmann coupling reactions | SpringerLink
DMSO-mediated palladium-catalyzed cyclization of two isothiocyanates via C–H sulfurization: a new route to 2-aminobenzothiazol

Figure 6 from Mechanistic study on the palladium(II)-catalyzed synthesis of 2,3-disubstituted indoles under aerobic conditions: anion effects and the development of a low-catalyst-loading process. | Semantic Scholar

Scheme 1 Synthesis of the chelating ligand 1 starting from commercially... | Download Scientific Diagram

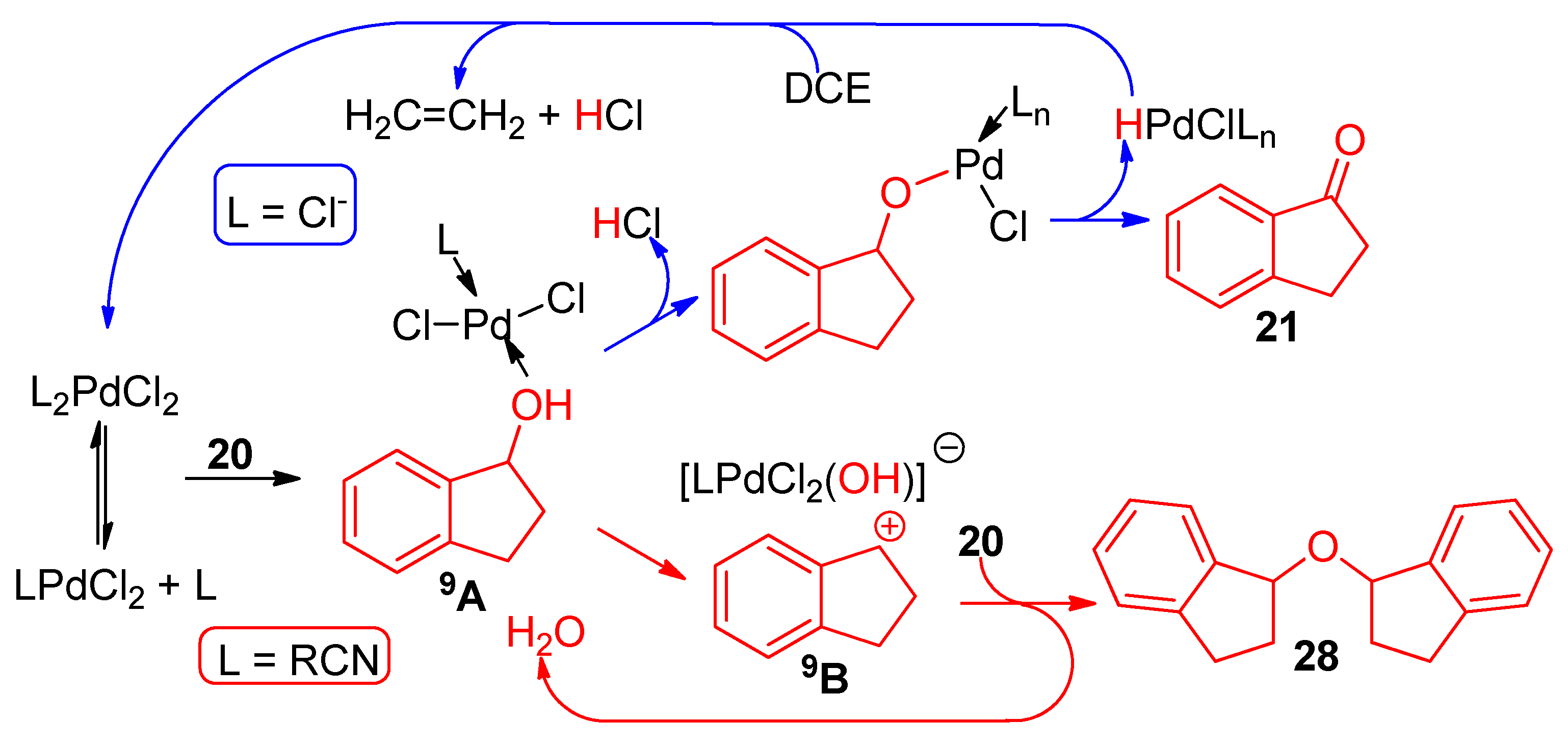
Mild aromatic palladium-catalyzed protodecarboxylation: kinetic assessment of the decarboxylative palladation and the protodepalladation steps. - Abstract - Europe PMC
Aqueous-Phase Chemistry of η3-Allylpalladium(II) Complexes with Sulfonated N-Heterocyclic Carbene Ligands: Solvent Effects in t

PDF) Reaction-Activated Palladium Catalyst for Dehydrogenation of Substituted Cyclohexanones to Phenols and H2 without Oxidants and Hydrogen Acceptors

Mild aromatic palladium-catalyzed protodecarboxylation: kinetic assessment of the decarboxylative palladation and the protodepalladation steps. - Abstract - Europe PMC
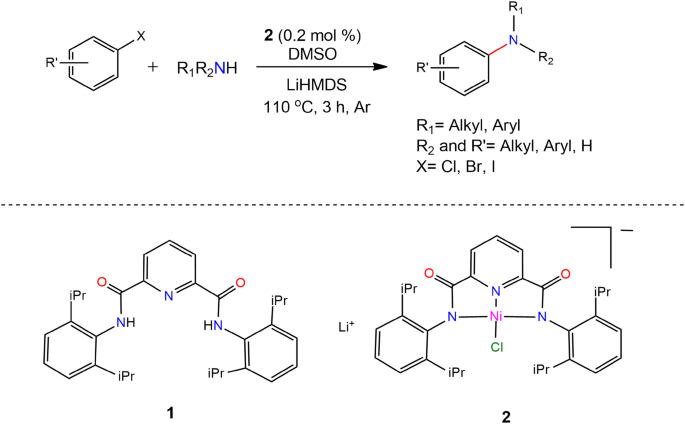
C–N Cross-coupling Reactions of Amines with Aryl Halides Using Amide-Based Pincer Nickel(II) Catalyst | SpringerLink
DMSO-mediated palladium-catalyzed cyclization of two isothiocyanates via C–H sulfurization: a new route to 2-aminobenzothiazol
![Synthesis and characterization of Pd based on [2,2'- bipyridin]-4-amine functionalized nano cellulose as a novel and recyclable nano catalyst for Suzuki reaction - ScienceDirect Synthesis and characterization of Pd based on [2,2'- bipyridin]-4-amine functionalized nano cellulose as a novel and recyclable nano catalyst for Suzuki reaction - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S1387700320305839-ga1.jpg)
Synthesis and characterization of Pd based on [2,2'- bipyridin]-4-amine functionalized nano cellulose as a novel and recyclable nano catalyst for Suzuki reaction - ScienceDirect